(public domain image)
When I first heard about the idea that radioactive decay might vary from the smooth, constant-half-life behavior that is typically observed, I was more than a little skeptical. As a nuclear chemist, I am well aware of how much energy it takes to affect nuclear processes. Since those energies are not generally attainable except with the use of a particle accelerator, a magnetic containment system, or some other high-powered device, it seemed absurd to think that variable radioactive decay was anything other than the mad wish of those who didn’t like the conclusions of radiometric dating. However, over the years, the data have convinced me otherwise. I written a couple of posts about variable radioactive decay (see here and here), and it seems clear to me that it does happen, at least under some circumstances.
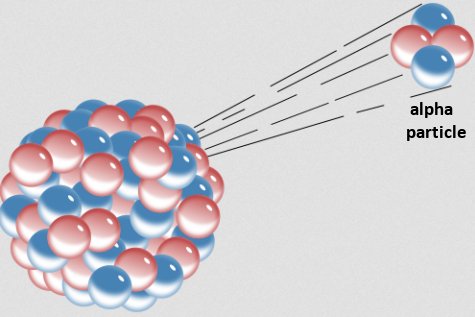
Recently, I came across another study on variable radioactive decay. It is actually a follow-up to a previous study,1, and it explores the alpha decay of uranium-232. As shown in the drawing above, alpha decay is one specific type of radioactive decay in which an unstable nucleus attempts to reach stability by spitting out two protons and two neutrons. Those four particles are bound together to form the nucleus of a helium atom, which for historical reasons is also called an alpha particle. It turns out that when uranium-232 does this, the resulting nucleus still isn’t stable, so a long series of further alpha decays occur, eventually producing lead-208, which is stable.
The authors of the study I am writing about weren’t interested in the subsequent decays. They looked specifically at the alpha decay of uranium-232. Under normal circumstances, this decay has a half-life of 69 years.* This means if I start with 200 uranium-232 atoms, after 69 years, only half of them (100) will remain. The other half will have decayed away. If I wait another 69 years, only half of those (50) will remain. In another 69 years, half of those (25) will remain. In the end, this is typically how radioactive decay works: the number of radioactive atoms ends up decreasing by half over every half-life.
The results of the study seem to indicate that a tabletop device involving a laser and gold can end up decreasing the half-life of uranium-232 by as much as a factor of 435,494,880,000,000!2
What did the authors do to obtain such an incredible result? They took a solution that contained dissolved uranium-232 ions and measured the number of alpha particles coming specifically from the decay of uranium-232. This is called the activity of the solution. They separated the solution into several samples and placed a gold target into each sample. They then fired one of four different lasers at the gold in each sample. After exposing the gold to the light from the laser for a while, they measured the activity of the sample. In each case, the activity was lower than it was prior to the laser burst, and the decrease was more than expected based on just the half-life of uranium-232.
Interestingly enough, however, the decrease in activity was different for each type of laser used. A copper vapor laser with a pulse that lasts for 20 billionths of a second produced the smallest decrease in activity, while a Nd:YAG laser with a pulse that lasted only 150 trillionths of a second produced the largest decrease in activity. Over the course of an hour, the most effective laser decreased the activity of the solution by a factor of two. Normally, it would take 69 years for the activity to decrease by a factor of two. With the right laser, however, it happened in just an hour.
What do the authors think happened to produce this dramatic decrease in activity? They think that when the laser hit the gold, it produced tiny particles (nanoparticles) of gold that dispersed in the solution. As the laser light continued to pulse, it interacted with those nanoparticles, producing a plasma effect that concentrated a large amount of energy into a tiny volume. This concentrated energy was enough to affect the process by which alpha decay occurs, speeding it up dramatically.
How dramatically did the decay speed up? According to the authors, during the hour over which the solution was exposed to the laser, the light was only hitting the gold for a very short amount of time. This is because the laser pulses, and each pulse is only 150 trillionths of a second long. In between each pulse, there is “dead time,” where no laser light is being generated. Based on the way this particular laser worked, the “dead time” lasted much, much longer than the laser pulses. As a result, the light was only hitting the gold for a grand total of 5 millionths of a second during the hour over which the sample was exposed. If the laser light really is what caused the decrease in activity, then, those 5 millionths of a second of light resulted in the equivalent of a full half-life of radioactive decay. Thus, the authors contend that the laser reduced the half-life of uranium-232 from 69 years to 5 millionths of a second!
Do I believe the author’s results? I am not really sure. I think the most convincing part of the analysis is that each laser produced a different decrease in activity. That seems to indicate there is something about the laser light that is causing the uranium-232 to decay faster than it should. Based on this and the earlier study, however, it seems that the laser light is not enough. There has to be a metal, like gold, involved as well. Once again, that seems to point to the fact that there is a real effect going on here.
At the same time, however, I have my doubts about the methodology. It seems to me they didn’t test this as much as they could have. According to the paper, the samples were exposed to the laser for only an hour. Why? It seems to me that they had the whole thing set up already. Why didn’t they expose the samples to the laser for an hour, measure the activity, and then do it for another hour? If the effect is real, they should see a further reduction in activity. Indeed, if each hour under the laser truly did produce a half-life worth of decay, they should have been able to reduce the sample to no detectable activity after about 10 such exposures. I contacted the lead author to ask why this wasn’t done, and I got no response.
Now don’t take this criticism in the wrong way. Overall, the data really do seem to indicate that this is a real effect. I hope someone else with the equipment can try to replicate this experiment and perhaps do several cycles of exposure to see how reproducible the effect is. Until that happens, I think the best I can say is that this is an interesting result that is worth investigating further.
*NOTE: The original version of this article incorrectly said the half-life was 69 hours. An astute reader informed me that I had the time unit wrong. 69 years is the correct half-life. The factor by which the half-life was decreased has been fixed as well.
Return to Text
REFERENCES
1. A. V. Simakin and G. A. Shafeev, “Accelerated alpha decay under laser exposure of metallic nanoparticles in aqueous solutions of uranium salt,” Physics of Wave Phenomena 19(1):30-38, 2011
Return to Text
2. A. V. Simakin and G. A. Shafeev, “Accelerated alpha-decay of uranium isotopes induced by exposure of aqueous solution of uranium salt with gold nanoparticles to laser radiation,” Physics of Wave Phenomena 21(1):31-37, 2013
Return to Text

Do you think a process similar to this could be used to dispose of radioactive waste?
I would think it would be an expensive process, Aaron, so I am not sure how practical it would be. However, if the effect is real, it most certainly could be used to dispose of radioactive waste.
Hi Dr. Wile – I’ve enjoyed teaching homeschool co-op classes using your science books. Thank you very much for writing them.
A couple of comments about this experiment –
I note that they don’t make any mention of measuring the activity of the solution during the 1 hour period of laser bombardment. Wouldn’t you expect it to go up dramatically during that time, to account for it going down later? How come no measurement of activity during the bombardment?
Given the above, is it possible that diffused gold in the solution is somehow blocking some of the alpha particles from being emitted from the solution, thereby giving a false activity reading?
If those two concerns I just mentioned were resolved, then I would be pretty impressed that these people are on to something.
If this effect is real, this has HUGE implications for nuclear physics. It would overturn a lot of assumptions about the amount of energy needed to affect nuclear reactions, and give new credibility to “cold fusion” theories.
Aren’t atomic clocks currently the basis for our time standard? I wonder what it does to our time standard if we discover that nuclear decay rates can be affected by the environment.
Thanks for your comment, David. Because of the way alpha particles are detected, the authors could not measure alpha particles during the laser bombardment. However, the authors did measure the gamma rays coming from the solution, and they did see a distortion in the spectrum when the laser was active. Now gamma rays aren’t a really good probe of alpha decay, but they are sensitive to the energetics of the decaying nucleus. Thus, it does seem the laser did something to affect the energetics of the process.
I doubt that the diffused gold particles can be responsible for the magnitude of the effect seen. I don’t know the exact concentrations, but nanoparticles don’t stay in solution at high concentrations. To block half of the alpha particles would required a lot of suspended gold – more than I would think the solution could contain.
I am not sure that this effect, if real, has huge implications for nuclear physics. In fact, the authors’ proposed mechanism is consistent with quantum mechanics as we understand it today, and it doesn’t require enormous changes in the energetics of the decaying nucleus. The authors discuss that there is an energy barrier that works against alpha decay, but by accepted mechanisms of quantum mechanics, the alpha particles can tunnel through that barrier. As they discuss, small changes to the barrier can have dramatic effects on the rate of tunneling. As a result, the issue isn’t really how much energy is involved. The issue is how that energy is absorbed by the nucleus.
Also, this would have nothing to do with cold fusion. Fusion is the process by which small nuclei are forced together to make a bigger nucleus. Radioactive decay is the process by which an unstable (usually large) nucleus spits out bits of itself to reach stability. The two processes are utterly different, and it’s not clear how a stimulus that affects one will necessarily affect the other.
Atomic clocks are the basis of modern timekeeping, but they have nothing to do with radioactive decay. Atomic clocks utilize the electromagnetic radiation emitted by excited electrons in an atom. This is an atomic effect, not a nuclear one, and it is strongly dependent on environmental factors. Because of this, the atoms in the most precise atomic clocks are cooled to near absolute zero and probed in a microwave cavity.
Dr. Wile, thank you for your answer and your patient explanations. I like how your response added to my knowledge. You are a great teacher.
Have you got any advice for a 46-year-old beginner who wants to understand quantum mechanics? I feel like my life is incomplete until I understand it better. I am a curious person who wants to know how everything works. And, as you point out in your textbooks, understanding the universe around us can bring us closer to God.
I have an education in electrical engineering and have worked for a couple decades as a software developer. I am re-reading Einstein’s book “Relativity: The Special and the General Theory – A Clear Explanation that Anyone Can Understand” for about the 4th time. When I get through with that, I intend to take another run at understanding quantum mechanics. Is there a book you would recommend to me?
Thanks,
David H
I am happy to help, David. I appreciate you taking the time to comment. I suspect that my readers have been further educated as a result.
Quantum mechanics is a strange discipline, indeed. However, because it works so incredibly well, it is definitely worth learning. I personally think the best starting point is The Quantum World: Quantum Physics for Everyone by Kenneth Ford. He has a companion book called 101 Quantum Questions. While there is some overlap, it is also a good book if you have time.