Not too long ago, a reader sent me an email that said:
I just found out about CRISPR and Cas9. From what I have learned about them they are very powerful and will lead to great things (good and terrible alike).
I was wondering if you could write a blog article on them when you get a chance. I would love to hear your perspective.
The reader is exactly right. CRISPR and Cas9 team up to make a powerful gene-editing tool that has incredible potential. While much of that potential is positive, some of it is quite negative.
To best understand the good and the bad of CRISPR and Cas9, you need to know what they are and what they can do together. CRISPR stands for “clustered regular interspaced short palindromic repeats.” Originally discovered in bacteria, it is a strand of RNA that is hooked to a CRISPR-associated protein, called a “Cas.” Cas9 is just one possible protein that can be associated with CRISPR, but it is the one that is most commonly used.
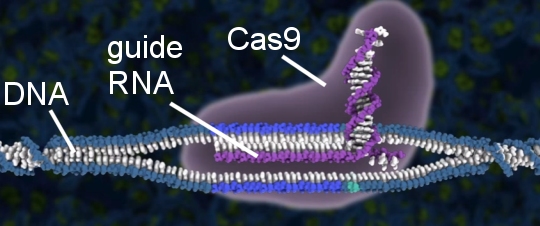
If your eyes are already glazing over, stick with me, because it’s important to understand how this works. RNA is a molecule that links to DNA. A specific RNA molecule targets a specific sequence of DNA. So, if you construct an RNA molecule correctly, it can search an entire DNA molecule, looking for a specific DNA sequence. It can then attach itself to that sequence. That’s what the RNA strand on CRISPR does. It is sometimes called “guide RNA,” because it guides the Cas9 protein to a specific part of an organism’s DNA. Consider the following illustration:
(taken from the video posted at the end of this article)
In the illustration, the guide RNA has found the DNA sequence that it was made to target. Since Cas9 is attached to the guide RNA, it is now positioned at a specific place in the DNA molecule.
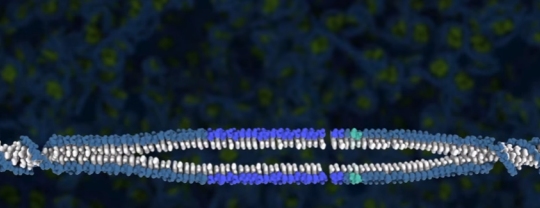
Once the Cas9 gets positioned at the proper place, it cuts the DNA molecule, removing a portion of it. This leaves the DNA molecule in two pieces:
(taken from the video posted at the end of this article)
If that’s the end of the story, the organism’s DNA repair mechanisms stitch the two parts of the DNA together, and the result is the organism’s DNA with that one section removed.
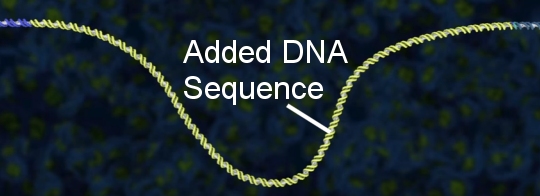
There is, however, another possibility. Once the DNA has been cut, a new DNA sequence can be spliced into the DNA right where the cut is:
(taken from the video posted at the end of this article)
So in the end, CRISPR and Cas9 can be used to either “edit out” a portion of an organism’s DNA, or replace a portion of the organism’s DNA with something completely different. Obviously, that’s a powerful tool! Also, it is fairly easy to make, so it is also convenient to use. No wonder the journal Science named it 2015’s scientific breakthrough of the year.
Now think about the potential. Suppose a scientist wants to figure out the function of a specific gene. He or she can cut a section of the gene out, rendering it nonfunctional. The scientist can then study the effect this has on the cell or organism being studied. But that’s just the beginning. Some genetic diseases are the result of a gene having one bad section. Remove that bad section (or replace it with a good section of DNA), and you get rid of the disease. In other words, CRISPR can be used to treat genetic diseases.
Indeed, this has already been done. Mice were engineered to have a mutation that causes Duchenne muscular dystrophy, a genetic disease that affects 1 in every 5,000 newborn male children. Most who get the disease end up being confined to a wheelchair by age 10 and die before they are 40. Scientists made a CRISPR-Cas9 system that targeted the mutated portion of the mouse DNA and cut it out. After injecting the system into the bloodstream of a mouse, the mouse showed significant improvement. Obviously, this treatment is a long, long way from being used in people, but the mouse experiment at least demonstrates that such genetic treatments are possible.
While both of these applications (editing out genes to see what they do and treating genetic disease) hold great potential, there are dangers that have to be considered. First, it is always possible for CRISPR-Cas9 to make a mistake and edit the wrong portion of DNA. This is typically referred to as an “off-target alteration.” Second, we actually know precious little about DNA. Indeed, just a few years ago, the scientific consensus was that most of the human genome consisted of “junk DNA.” We now know how spectacularly wrong this evolution-inspired idea was, but it serves to illustrate how little we really understand DNA. As a result, even when CRISPR-Cas9 does exactly what it is supposed to do, there could be unintended consequences.
Finally, the most important possible danger associated with CRISPR-Cas9 involves the specific cells in which the DNA editing occurs. If CRISPR-Cas9 is used to target the cells in most tissue (like muscle tissue, nerve tissue, etc.), the only consequences that exist are for the specific organism being treated. However, if it is used to target the cells involved in reproduction (typically called “germline tissue”), the result of the gene editing will be passed on to subsequent generations. Given the possibilities of off-target alterations and unintended consequences, this could devastate an entire lineage!
Because of these very real dangers, several scientists (including some involved in the invention of CRISPR-Cas9) have called for prudence and caution. In their statement, they write:
At present, the potential safety and efficacy issues arising from the use of this technology must be thoroughly investigated and understood before any attempts at human engineering are sanctioned, if ever, for clinical testing. (emphasis mine)
I wholeheartedly agree. This new gene-editing technology holds great potential, both for good and for bad. Scientists need to exercise extreme caution in how it is used.
If you would like to see a video of how CRISPR-Cas9 works, this one was posted by one of its co-inventors:

I wonder what the Biblical implications of this would be? It’s always hard to know whether or not we are supposed to be doing this stuff. Obviously God gave scientists the wherewithal and inclination to begin to understand his blueprints…. but how much we are allowed to meddle with these things? Very hard to take a position. It feels like the edits they’ve been making with GMO crops have been pocket book motivated. All its resulted in is more herbicide use.
I will say this… The strange beasts of revelation don’t seem that farfetched in these times.
If this technology progresses, in 100 years or so we may be able to treat the human body like a Word document.
What a terrifying and exciting possibility.