I am a nuclear chemist. I chose to be a nuclear chemist because studying the nucleus fascinates me. It’s amazing enough to study something we can never hope to actually see, but the fact that the nucleus is shrouded by clouds of electrons makes the job all the more fun.
Because I am a nuclear chemist, there are certain things I don’t want to believe. For example, I don’t want to believe quantum mechanics is wrong just because it is incompatible with general relativity. Don’t get me wrong, I don’t want general relativity to be wrong, either. Black holes, white holes, and wormholes are just far to fun to ever want to give up! Nevertheless, if I have to choose one of those two theories to be wrong (because they are incompatible), I will to choose general relativity, because quantum mechanics works so incredibly well when it is applied to small things like the nucleus.
For a long time, there was something else I didn’t want to believe. I didn’t want to believe that the half-lives of radioactive isotopes could change. It seemed so clear to me at the time: radioactive half-lives depend on the energetics of the nucleus, and the energy levels in the nucleus are (roughly speaking) about 100,000 times that of the electrons in an atom. Thus, if nature exposes a radioactive atom to stress, the electrons should be the ones that deal with the stress, not the nucleus. The nucleus is under the electron cloud, and it deals in energies that are so much greater than electron energies, that the electrons effectively “shield” the nucleus from being affected by most of the stress that nature can throw at it.
Over the years, however, the data have drug me (kicking and screaming the entire way) to the point where I have to admit that radioactive half-lives can change, and in some cases, they can change quite substantially.
For example, Otto Reifenschweiler showed that when tritium (a radioactive form of hydrogen) was incorporated into small particles of titanium and heated, the radioactivity of the tritium decreased by 40%1. This means the half-life of the radioactive decay increased. F. Raiola and his collaborators saw a roughly 6 percent decreased half-life for a radioactive form of polonium when it is layered in copper metal. 2. Interestingly enough, Raiola’s paper references another study that I am waiting on from interlibrary loan in which the same form of radioactive polonium had a 300% shorter half-life when it is layered in palladium. (NOTE: After reading the paper, I think Raiola and colleagues misinterpreted the results; they do not imply a 300% shorter half-life.)
Nuclear chemists and physicists have no coherent model to account for these changes in radioactive half-life. However, that is not unusual. It often takes generations for theorists to actually explain why experimentalists see what they have seen. Also, the explanation is not nearly as important to me as the fact that it clearly can happen, even though all my education (through graduate school) said that such a thing is clearly impossible.
In fact, it seems that what was considered impossible just 20 years ago is being demonstrated all over the place. Indeed, there is now clear and convincing evidence that the rate of radioactive decay varies (slightly, but still measurably) based on the distance between the earth and the sun! 3 Interestingly enough, two independent labs have been seeing this effect since the early 1980s, but both were too afraid to publish the results, thinking they were making some systematic error. Instead, the error is clearly in the assumption that radioactive half-lives are constant.
The most dramatic demonstration of the idea that radioactive half-lives are not necessarily constant, however, has been provided by young-earth creationists. Back in 1982, Robert Gentry was studying zircons in what is supposed to be Precambrian rock, which is supposedly 1.5 billion years old. 4 He found an enormous amount of helium trapped in those zircons, and that doesn’t make sense if radioactive half-lives were always the same.
You see, helium is a product of one type of radioactive decay – alpha decay. In this form of radioactive decay, a nucleus spits out two protons and two neutrons to become a completely different nucleus. Those two protons and two neutrons capture two free electrons, producing a helium atom. In the zircons studied, uranium and thorium were the nuclei that spit out alpha particles, making helium atoms. Well, in the lab, this kind of helium production is very slow, because the rate-determining half-lives for the alpha decay are very slow. Thus, if radioactive half-lives have always been the same, we can assume that the helium production in these zircons occurred very slowly over time.
So what happens to the helium that is produced? Well, it is a small atom, and it is unreactive. Over time, then, it simple “squeezes” through the spaces in between the ions that make up the zircon, escaping the zircon. This is much like the helium-filled latex balloons you buy from the store that stay inflated for only a few days. The helium inside the balloon actually “squeezes” in between the polymers that make up the latex, escaping the balloon.
The situation, then, is that helium is produced in the zircons, but then it escapes from the zircon. If radioactive half-lives have always been what they are right now, then the helium production in zircons is slow. It was always assumed (but not measured until recently) that the rate at which helium escapes from zircon is relatively fast. Thus, a slow rate of helium production coupled with a fast rate of helium escape means that there should not be any appreciable helium in the zircons Gentry was studying. However, there was. This indicated to Gentry (and some others) that helium must have been made significantly faster in the past. In fact, it was being made so fast that it could not escape quickly enough, and the amount of helium in the zircons “piled up” to the point where it is still trying to escape today.
To see if this reasoning was right, another young-earth creationist, D. Russell Humphreys, decided to find out if anyone knew the rate at which helium escaped from zircon. He found that it had never been measured. Therefore, he decided to get someone to measure the rate at which helium escapes from zircon. However, before the study was commissioned, he proposed two scenarios: 5
1. Assume that radioactive decay rates have always been the same and that the rocks are 1.5 billion years old, as assumed by old-earth geologists.
2. Assume that there was a “burst” of very fast radioactive decay about 6,000 years ago which produce a quick buildup of helium, and the helium has been escaping since then. During the “burst,” radioactive half-lives would have been VERY SHORT compared to what they are today.
What is nice about these two scenarios is that they each need a specific rate of helium escape in order to be true. If option (1) is true, the rate of helium escape must be very slow – slower than the rate at which helium is currently produced in zircons. If option (2) is true, the rate of helium escape must be much faster. Thus, each scenario provides us with a prediction for how quickly helium should be able to escape from zircon. It turns out that the helium escape rates predicted by these scenarios are different by more than a factor of 100,000.
So here we have two different predictions for the rate at which helium escapes zircon, and at the time the prediction was made, no one knew what that rate was. Humphreys then commissioned a group that didn’t know any of the details of this work to go into the lab and actually measure the helium escape rates. Guess what was found?
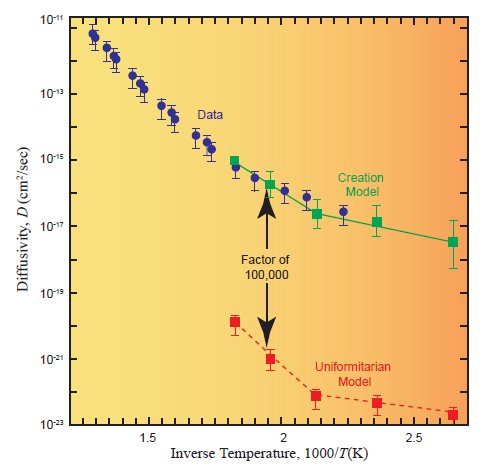
(taken directly from reference 5, page 55)
The red curve, labeled “Uniformitarian Model,” is option 1. The green curve, labeled “Creation Model,” is option 2. The blue dots with error bars are the data, measured by a group that didn’t know anything about either model. Note that the data say option 2 is right and option 1 is miserably wrong.
To me, this is very striking. After all, in the scientific method, you produce a hypothesis, and you then use that hypothesis to make predictions about the data. If the data fall in line with those predictions, there is evidence for the hypothesis. This is exactly what was done here. The hypothesis that radioactive half-lives have been constant for 1.5 billion years led to one prediction, and it fails miserably when compared to the data. The hypothesis that radioactive half-lives were significantly shorter a few thousand years ago led to a prediction that ended up being supported by the data, once the data were actually measured by an independent party.
As a result, even though I don’t like it, I have been drug, kicking and screaming the entire way, to the distasteful conclusion that radioactive half-lives are not constant. I don’t like it, but science isn’t something you necessarily like or don’t like. It is something that is determined by the data, regardless of how ugly it ends up being.
REFERENCES
1. O. Reifenschweiler, “Reduced Radioactivity of Tritium in small Titanium Particles”, Phys. Lett. A, 184:149 (1994).
Return to Text
2. F. Raiola, et. al., “First hint on a change of the 210Po alpha-decay half-life in the metal Cu”, Eur. Phys. J. A 32:51–53, 2007
Return to Text
3. Jere H. Jenkins, et. al., “Evidence for Correlations between Nuclear Decay Rates and Earth-Sun Distance,” arXiv preprint 0808.3283v1 [astro-ph], August 25, 2008. Available online
Return to Text
4. Gentry, R.V., et al., “Differential Helium Retention in Zircons: Implications for Nuclear Waste Management,” Geophysical Research Letters, 9:1129-1130, 1982
Return to Text
5. Young Helium Diffusion Age of Zircons Supports Accelerated Nuclear Decay
Return to Text

I see that the Gentry article is less than two pages. Can you reproduce it here? At least a link to it’s abstract would be helpful.
Reproducing it here would probably be a violation of copyright. However, I can type in the abstract:
“A very sensitive helium leak detector was utilized to measure the helium liberated from groups of zircons extracted from six deep granite cores. The observed low differential loss of gaseous helium down to 2900 m (197°C) in these ancient Precambrian rocks is easily attributable to the greater diffusion of He at higher temperatures rather than losses due to corrosion of the zircons. This fact strongly suggests that deep granite burial should be a very safe corrosion-resistant containment procedure for long-term waste encapsulation.”
Essentially, Gentry and his colleagues heated up the zircons and measured the amount of helium that leaked out. They continued to raise the temperature until no more helium came out.
Thanks, but I found it via the horse’s mouth: http://www.robertvgentry.com/
It seems Gentry was a nuclear physicist who published in peer-reviewed journals in the 1970s on radiohalos, had two articles on the zircons in 1982, then took some time off. He reemerged in 1997, now writing about astrophysics, specifically how Genesis supports the Big Bang theory, or the other way around, or doesn’t support it, I’m not really sure. Anyway, he doesn’t list any new credentials, but is affiliated with the Orion Foundation. Its website claims the suppression of his recent articles is “The Great 21st Century Scientific Watergate” and tells how his lawsuit to restore publication on arVix, the open source preprint database, was dismissed. arVix did change their policies because of the incident, however.
His career path reminded me of yours, except your field is now biology, not astrophysics. And you haven’t sued anybody to my knowledge.
I have no idea why you think my “field is now biology.” That’s nonsense. My current field is science education, which has ALWAYS been my field, because I have been teaching (and winning awards for it) since graduate school.
Sorry, I was thinking about our debate on evolution. Which Apologia texts have you authored?
I have authored
Exploring Creation with General Science
Exploring Creation with Physical Science
Exploring Creation with Chemistry
Exploring Creation with Physics
Advanced Chemistry in Creation
Advanced Physics in Creation
Since I am NOT a biologist, I needed a co-author for the two biology-related books on my list of credits:
Exploring Creation with Biology (co-author Marilyn Durnell)
The Human Body (co-author Marilyn Shannon)
Thanks. Do you have any education credentials?
I taught chemistry at the University of Rochester and Indiana University, and then I taught chemistry and physics at Ball State University. During my time as a science educator, I was awarded:
Excellence in Teaching Award (Ball State University)
Outstanding Teacher Award (University of Chicago)
W.D. Walters Award for Teaching Excellence (University of Rochester)
Westinghouse Science Talent Search Certificate of Honor (Science Service)
Not teaching awards, education credentials, such as an undergraduate minor in education, to set the bar low.
I think you need to learn the English language a bit better. You asked for credentials. According to the Merriam-Webster dictionary, credentials are:
1 : something that gives a title to credit or confidence; also : qualification
2 : testimonials or certified documents showing that a person is entitled to credit or has a right to exercise official power
The fact that I have taught at three universities obviously gives title and confidence to my role as a science educator. In addition, the awards are “certified documents” showing that I have a “right to exercise” my role as a science educator. In fact, they show that I do it in an exemplary way.
Also, perhaps you are too uneducated to understand that professors do not have education degrees. Nevertheless, professors teach those who are getting their education degrees. Being a professor, then, is a significantly higher education credential than an education degree.
I’ll take that as a no. While you have your dictionary open, why don’t you look up “pedagogy.” I am educated enough to know that being a professor is very different from writing a textbook. Most textbook writers have education credentials, such as a PhD in Educational Psychology. In fact, for younger students, since the material isn’t exactly cutting edge research, educational background is much more relevant than subject matter expertise. Are there any textbooks in use in Indiana public schools whose authors don’t have degrees in education?
You are continuing to show your ignorance by claiming that the majority of textbook authors have education degrees. They certainly do not, at least when it comes to the sciences. For example, the most popular biology textbook in Indiana is “Modern Biology,” published by Holt, Rinehart, and Winston. The book lists 10 contributing writers (along with the original author, Albert Towle), and NONE has an education degree. All the degrees are either PhDs or MSs in a biology discipline. There are two who have no degrees but have spent years writing about science.
Sticking with that same publisher, their high school physics text, “Physics,” is one of the most popular high school physics texts in the country. Its two authors, Raymond A. Serway and Jerry S. Faughn, have PhDs in physics, and neither has any education degrees.
What all these authors (and indeed, the majority of science textbook authors) have in common, however, are serious education CREDENTIALS. They are all either professors or (as is the case with two of the contributors to the biology book mentioned above) have years of experience educating people about science through the written word. Once again, if you learned enough English to understand what “credentials” are, you would not embarrass yourself like this.
By the way, if you bothered to educate yourself on pedagogy, you would find that it is defined as:
the art, science, or PROFESSION of teaching
Thus, as anyone with a modicum of understanding of education knows, any experienced teacher is very well-versed in pedagogy. One reason my textbooks are so wildly popular in both homeschools and accredited Christian schools in the US and Canada is that I know pedagogy so well. As a result, students can really learn from my texts. This is easily demonstrated by the fact that so many students who use my texts end up being such excellent university science students.
I hope this teaches you to educate yourself on issues before you start making pronouncements about them. When you make silly statements that are so easily refuted, you just end up embarrassing yourself.
Thanks for the smack down. I appreciate it. While I have enjoyed this digression from Gentry into science textbooks, I have forgotten to post the reply to Humphreys’ work.
http://www.talkorigins.org/faqs/helium/zircons.html
No problem. Always happy to properly put in their place people who try to challenge credentials rather than reply with substance. Next time, check your facts so that you don’t embarrass yourself.
Speaking of ignoring substance, Humphreys has already thoroughly refuted the baseless nonsense on talkorigins. If you are interested in being educated on this issue, you can find his replies here:
http://www.icr.org/pdf/rate/humphreys_to_hanke.pdf
http://www.trueorigin.org/helium02.asp
http://creation.com/helium-evidence-for-a-young-world-continues-to-confound-critics
Even though the talkorigins site references the first two responses, they don’t reference the last one. I find that very interesting.
As you can see, then, even an incredibly deceptive site like talkorigins is unable to distract from what the data really say.
I read Henke’s article, so I saw Humphreys’ replies, and I thought he refuted nothing. For example, did he refute this:
“What Dr. Humphreys’ data really say. Using the best available data, the “creation dates” from the equations in Humphreys et al. (2003a) provide ridiculous answers that range from hundred to millions of “years” (an average and two standard deviations of 60,000 ± 400,000 “years” old, using only one significant digit). Because Dr. Humphreys’ helium diffusion data were obtained under a vacuum rather than under pressures that realistically model the subsurface conditions at Fenton Hill, the actual helium diffusion results at Fenton Hill are probably orders of magnitude lower as illustrated by the blue arrows.”
No, instead he says things like: “To our great delight, the data fell right on the ‘6,000 year’ prediction!” and “The close fit of the model and the experiment is strong evidence that both are essentially correct, because the probability of an accidental fit is low.” It would be much more honest to put the words “data” and “model” in YEC papers in scare quotes.
By interesting, do you mean that since Humphreys posted last, he wins? That is interesting.
From the creation.com link: “None of the critics listed below have published their denunciations in peer-reviewed scientific publications. Instead they are ‘lone-ranger’ opinions in un-reviewed venues such as Internet sites and seminars. This contrasts starkly with the RATE helium project. It was a multi-author effort, and it had more than seventeen reviewers and editors as it appeared in five technical publications, one of which is non-creationist”
As Henke stated, it would be hard to justify including a refutation in a peer-reviewed scientific publication of a paper that didn’t appear in a peer-reviewed scientific publication to begin with. No, the ICR and CRSQ don’t qualify. What scientific journal database includes these sources? I can’t find them on Academic Search Premiere, or any other electronic journal source.
Humphreys claims that many authors beat one author. Is that your idea of scientific reasoning?
It’s really interesting Humphreys calls his sources “technical,” not “peer-reviewed scientific” publications. Then he says one is non-creationist. (Nice to admit 4 are creationist) But that non-creationist source is not a publication, but rather a poster at the AGU meeting in 2003. I’m wondering, has the AGU invited Dr. Humphreys back to present any of his work?
But absolutely the most interesting thing about the creation.com link was that all the critics listed except Dr. Henke were fellow creationists. Argue all you want over YE or OE, but that doesn’t say anything about whether creationism has any scientific validity.
You obviously don’t understand the physics involved here, as Humphreys clearly did refute the nonsense written by Henke. Of course Humphreys refuted the vacuum argument, as anyone who understands basic physics would see. As Humphreys clearly says:
“However I have at hand a paper [Carroll, M. R. 1991. Diffusion of Ar in rhyolite, orthoclase, and albite composition glasses. Earth and Planetary Science Letters 103:156-168] that gives, among other data, the pressure effect on argon diffusion in glasses, such as rhyolite obsidian. At the highest temperature to which our helium-in-zircon experiment went, 500 degrees C, the pressure effect on the glasses was almost imperceptible, a few percent per kilobar.”
This is VERY similar to the situation in Humphreys’s experiments. Thus, data measured in a very similar situation show that the effect is insignificant. Humphreys then goes on to give three very physically accurate reasons why the effect in his experiments would be less than one percent. This is a complete refutation of Henke’s nonsense.
It is much more accurate to put Henke’s “refutation” in scare quotes.
By interesting I mean that the talkorigins site makes it look like Humphreys did not respond again, when he clearly did. This is just more evidence for the incredibly deceptive nature of the talkorigins site.
Humphreys’s claim is not that many scientists beat one scientist. It’s that he had the guts to submit his research to peer review, but Henke does not. You can claim that the peer-reviewed journals that Humphreys published in “don’t qualify,” but once again, you are embarrassing yourself by making statements from ignorance. I have published in the peer-reviewed journals of the creationist community, and their peer review is significantly more rigorous than that of the secular peer-reviewed journals in which I have published. In addition, I have been a peer reviewer for both, and my reviews were handled more critically by the editorial staff of the creationist journals than they were by the secular journals. Thus, the journals in which Humphreys published do, indeed, qualify – better than the secular journals in which I have published.
You further show your ignorance by somehow implying that a poster session is not a publication. It certainly is, as it is a public display of the data that is intended for other scientists. You also don’t seem to understand how publications work, as that kind of meeting doesn’t INVITE speakers. You SUBMIT A PROPOSAL for a session, and that proposal is either accepted or rejected. In order to submit the proposal, you need to have a new, novel result. To my knowledge, there has been no new, novel result in this research since the poster was presented in 2003. The only progress is that it has withstood the attacks of many critics.
I agree with you that it is very interesting that the majority of critics of Humphreys are creationists. It shows how scientific creationism works. Real science is done when scientists CRITIQUE other scientists’ work. This allows the bias of one investigator to be removed from the analysis of the data. Unlike Henke, Humphreys is eager to have his work evaluated. As he says in his conclusion, this makes him even more confident of the results.
This is exactly what I thought when I first read Henke’s work four years ago. Henke’s analysis was so poor and so easily refuted (even before Humphreys had a chance to reply, I saw that Henke’s vacuum argument was absurd) that I became even more confident in the data. If someone who obviously didn’t want to accept the results couldn’t come up with any better arguments against them, then the results are clearly very reliable.
“No, the ICR and CRSQ don’t qualify. What scientific journal database includes these sources? I can’t find them on Academic Search Premiere, or any other electronic journal source.”
Bueller? Bueller?
Since you seem so ignorant about the English language, let me define peer review for you. According the the Microsoft Encarta Dictionary, it is:
an assessment of an article, piece of work, or research by people who are experts on the subject
Do you see ANYTHING in there about being included in some group’s database? Of course not. A peer-reviewed journal need not be carried in some database. It needs to be a journal in which articles are reviewed by experts on the subject. Could I perhaps send you a dictionary so I don’t have continue to teach remedial English to you?
Since you know nothing of the peer review process or of the specific peer reveiwers in these journals, you are not only showing your ignorance of the English language, but also of what makes a good peer-reviewed journal. As someone who has both published in and peer reviewed for them, I know. The peer review in those journals is significantly more rigorous than what happens in most other journals. You are continuing to show your ignorance by claiming something that is simply not true.
I would also point out that the absurd statement, “it would be hard to justify including a refutation in a peer-reviewed scientific publication of a paper that didn’t appear in a peer-reviewed scientific publication to begin with” shows Henke’s (and your) incredible ignorance concerning the scientific literature. In fact, peer-reviewed journals publish articles that attack ideas published in non-peer-reviewed forums ALL THE TIME. For example, Nature Reviews Microbiology (vol 4, pp. 784-790, 2006) published an article entitled “From The Origin of Species to the origin of bacterial flagella” by MJ Pallen and NJ Matzke. Here is the abstract:
“In the recent Dover trial, and elsewhere, the ‘Intelligent Design’ movement has championed the bacterial flagellum as an irreducibly complex system that, it is claimed, could not have evolved through natural selection. Here we explore the arguments in favour of viewing bacterial flagella as evolved, rather than designed, entities. We dismiss the need for any great conceptual leaps in creating a model of flagellar evolution and speculate as to how an experimental programme focused on this topic might look.”
This article, published in a peer-reviewed journal (probably not as rigorous as CRSQ, but still peer-reviewed), is responding to statements made IN A TRIAL and in NON-PEER-REVIEWED BOOKS. This happens all the time in the scientific literature. Thus, EVEN IF Humphreys’s work hadn’t been published in peer-reviewed journals (it was – but even if it hadn’t been), it would still be fair game for attack in peer-reviewed journals. The fact that neither you nor Henke seem to know anything about this just parades your ignorance of the scientific literature to the entire world.
Once again, educate yourself on issues before making pronouncements on them. That way, you don’t embarrass yourself so much!
Kicking and screaming, so apropos.
Indeed. You are kicking and screaming against the obvious conclusions of the data. The problem is, you don’t have any arguments to marshal against them, so you try to argue credentials and whether or not a journal is peer-reviewed. However, you can’t even get THOSE arguments right. I can see why you are so frustrated!