Let me start by saying that I have no connection to Pfizer or any other pharmaceutical company. I am a science educator who writes about science issues like this one. I am also not a medical doctor or medical researcher. I am simply a nuclear chemist who has broadened my knowledge base by writing (or co-writing) a series of textbooks used by home educators and teachers in Christian schools. Thus, I am no expert on these matters. However, I get most of my information by reading the scientific literature, which allows me to avoid a lot of the misinformation found in the standard media outlets and (even worse) social media.
Before I talk about Pfizer’s vaccine in particular, I want to explain how this kind of vaccine works. To understand that, remember that a traditional vaccine uses a weakened/inactivated form of the pathogen whose infection it wants to prevent (or a chemical mimic of that pathogen). This causes your body to react as if it is being infected by the real thing. As a result, it mounts a defense that is specific to that pathogen and remembers how to fight it. That way, if you get infected by the real thing, it can mount a swift immune response. This process takes advantage of your acquired immune system. However, you also have an innate immune system, and the active ingredient of the vaccine does not stimulate it. As a result, traditional vaccines have additives, called adjuvants, which are designed to stimulate your innate immune system. That way, everything in your immune system works the way it is designed to work.
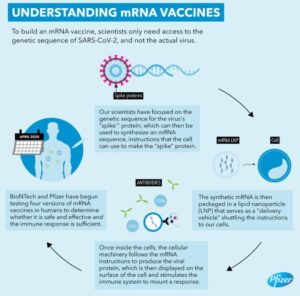
This new kind of vaccine takes a different approach. Rather than injecting you with a weakened/inactive version of the pathogen (or a chemical mimic), it injects you with the instructions that your cells need in order to make a protein that is found on the pathogen. These instructions are coded in a molecule called messenger RNA (mRNA), which is the way a cell expects to receive such instructions. Some of the cells in your body will make the protein according to those instructions and “display” it, which will cause your immune system to think it is being attacked by a pathogen. This will stimulate your acquired immune system to respond. It will then remember that response so if you get attacked by the real thing, it is ready.
You can’t just inject mRNA into the bloodstream, however, as it will be removed rather quickly. Thus, the mRNA is enclosed in a fatty membrane called a “lipid nanoparticle.” The membrane and its mRNA can enter your body’s cells. Once that happens, cells recognize the mRNA as instructions for making a protein, and it is sent to a protein-making center called a “ribosome.” Interestingly enough, the membrane-enclosed RNA also stimulates the innate immune system, so this kind of vaccine needs no adjuvants, at least based on what has been seen so far.
But what happens after the mRNA is used at the ribosome? After a while, it decays away and is recycled by the cell. Why? The DNA in the nucleus of each cell contains instructions for making the proteins that the cell needs to make, but it never leaves the nucleus. As a result, the instructions are sent from the nucleus in the form of mRNA. Thus, there is a lot of mRNA in the cell, and all used mRNA decays and is recycled as a matter of course. This kind of vaccine just adds one more mRNA to the cell, and the cell treats it like it treats any other mRNA.
But wait a minute. If mRNA comes from DNA, is it possible that this mRNA can somehow interact with your DNA, fundamentally changing it? Not based on what we know about the cell. DNA is found in the cell’s nucleus, and mRNA cannot enter the nucleus; it can only leave the nucleus. Thus, there is no way this mRNA can affect your DNA. There is a class of viruses called “RNA retroviruses” that have the ability to take their RNA and convert it into DNA, which can then enter the nucleus. However, that virus has a special chemical (a reverse transcriptase enzyme) and other support chemicals to get that job done. With this kind of vaccine, there aren’t any chemicals like that, so as far as we know, there is no way for this mRNA to even come close to the cell’s DNA.
Why would you want a vaccine like this? Because it has several advantages over a traditional vaccine. First, there are no adjuvants. Every time you add another chemical to anything that goes in the body, you have increased the possibility of side effects. Fewer chemicals should mean fewer possible side effects. Second, since this is fully synthetic, it is relatively fast to produce. Thus, it is thought that this kind of vaccine can be made for situations just like this one, where the sudden introduction of a pathogen produces a new disease that is infectious. Third, the development of the vaccine doesn’t require the virus itself. All you need is the sequenced genome. Thus, there no chance of a virus-related accident during production.
Regarding Pfizer’s version of this vaccine, not much is known. The early clinical trial has been published, and its results are very encouraging. Only 45 volunteers were involved. 36 got the vaccine, and 9 got a placebo. Those who got the vaccine produced more antibodies than what is seen in patients who have recovered from COVID-19, indicating that the vaccine produces a more than adequate immune response. Almost all of the volunteers who got the vaccine reported pain at the injection site, and many reported headache, fatigue, and fever, which were dependent on the dose that was given. Only 2 of the 9 placebo-injected people reported such side effects. There were no severe side effects for anyone. A later clinical trial involved 195 participants and two versions of the vaccine. One version produced fewer side effects than the other, but the immune responses in the participants were similar to those in the first trial. Once again, then, very encouraging results.
Of course, both clinical trials had small test groups, which is why the current clinical trial (for the version that produced fewer side effects in the second trial) has 43,538 participants. Unfortunately, the data related to that trial are not available, since it is not over yet. However, based on the information I linked previously, that trial has reached a milestone: 94 patients have tested positive. This is important, because 94 infections among the participants is statistically significant enough to conclude whether or not the vaccine is protective against the virus. Based on what the company is saying, more than 90% of those cases must have been in the group that received the placebo, which means the vaccine provides strong protection against the disease. However, we don’t know that yet, since the data have not been released.
Nevertheless, Pfizer cannot get emergency authorization for this vaccine until the trial has been going for two months, because the side effects must be tracked for at least that long. Supposedly, that milestone will be reached by the third week of this month. The clinical study will continue until at least 164 of the participants have been infected so that a better estimate of the vaccine’s effectiveness can be determined. I am personally very excited about this vaccine (and other candidates like it), but until more data are published, I will withhold judgement. However, if the data from the current clinical trial are as good as the data from the early one, I will be eager to get an injection.
I will add one note of caution. This kind of vaccine has never been used before, except in clinical trials. Thus, nothing is known about its long-term effects, except what is predicted based on our current knowledge of cellular processes. Those who are more cautious about medical interventions should take that into account. Of course, I must also add that we don’t know what the long-term effects of COVID-19 infections are either, so one must weigh both unknowns when making any decision related to the disease.
I will keep my readers apprised of any new developments.
NOTE (added 11/17/2020): Pfizer’s competitor on this kind of vaccine is a company called Moderna. In response to Pfizer’s press release, they released one of their own. It indicates similar efficacy and safety, which is great news. Moderna’s vaccine follows the same methodology as Pfizer’s and is also currently in a large clinical trial. The major difference between the two vaccines is that Pfizer’s requires specialized ultra-cold storage (-80 degrees C), while Moderna’s can be stored in any standard freezer (-20 degrees C). Pfizer’s vaccine will keep for 5 days in a refrigerator, while Moderna’s will keep for 30 days. As a result, it is thought that Moderna’s vaccine will be easier to distribute, if it gets authorized for use.

Thank you for sharing this detailed information! So interesting to learn about the new technologies being developed. It reminds me just how intricately God created things and how we still have so much more to learn.
I’m a little fuzzy on innate vs acquired immunity, but since this stimulates the innate immunity, does that mean that even after that mRNA decays, the body will always ‘remember’ how to fight this? Or would you need to re-teach the cells every so often?
Also, I’m curious how this might positively impact the development of future flu vaccines. Would be interesting if this same type of solution could be applied there.
I really appreciate your balanced approach to the topic of vaccines: weigh the risks/unknowns of the vaccine against the disease. Unfortunately, at this point we don’t know the long-term effects of either.
Innate immunity is a general defense against any infection. The innate immune response triggers some aspects of the acquired immune response, which is why both should be triggered for maximum effect. The acquired immune system is what remembers the infection. It makes cells called memory B-cells and memory T-cells, which are ready to start the fight right away. So as long as the virus stays the same, the acquired immune system’s memory cells should remember the infection so that there is no need for retraining.
This kind of vaccine could be used for the flu, which would probably get rid of the need for adjuvants. However, the problem with the flu vaccine is that the flu strains keep changing, and the memory cells aren’t as good as fighting different strains. They offer some protection, but the more different the strain becomes, the less protection they offer. This kind of vaccine wouldn’t offer any improvement in that regard.
You didn’t answer the million dollar question. Will you take it and if so how long would you wait?
It’s so hard to know when to embrace a new technological advancement when you look back and see these instances where it took decades to realize the long term harm imparted.
As I said in the post, if the large trial results are as good as the small trial results, I will eagerly take it. I agree that it is a risk, but not taking it is a risk as well. I think the later risk is greater than the former, especially for someone my age.
Dr. Wile,
Thanks for writing this article; I now have a better understanding of how the mRNA vaccine works. I’m curious if you have done any research on fetal cell lines that are used in vaccines, and if so, are they using them in this vaccine? Also, have you done any research on digital tracking or tattoos that may be injected under the skin with this vaccine? The aborted fetal cell lines are obviously a problem from a biblical and moral standpoint, but digital tracking and tattoos are concerning as well from the standpoint of our civil liberties. If true, these would also condition the world for the mark of the beast that will happen during the tribulation which we do not want to promote.
Here are some articles I have read recently:
https://greatgameindia.com/invisible-tattoo-coronavirus-vaccine/
https://www.prophecynewswatch.com/article.cfm?recent_news_id=4391
Hi Heath,
I have written about the fact that a few vaccines use cell lines from abortions that took place decades ago. I agree with nearly every pro-life organization on the planet that people should use them, because in the end, they do save lives. I don’t see how protesting abortions that took place decades ago is worth sacrificing precious human lives today. However, I can see why some pro-lifers hesitate. The important thing to realize is that it applies to only a few vaccines, and use of those vaccines doesn’t increase the number of abortions done, since the cell lines are self-perpetuating.
Vaccines like this one do not grow viruses, so there is no need for host cells. As a result, there is nothing related to fetal tissue in the development of these vaccines.
The dye being developed at MIT cannot be in any vaccine right now (including the COVID-19 vaccines), because it hasn’t gone through the clinical trials necessary to authorize its use. Also, as you can see from the article, it requires a special delivery device that would have to be separate from the vaccine injection. Thus, this could become an issue later on, but it is not an issue right now.
As far as the “COVID passport” issue, that’s a political one, and such discussions tend to produce more heat than light. I will not comment further on such issues.
Oh I missed that somehow! I see it now. Scares me but like you said, risk exists either way. I’d love to hear Dr. Sanford’s take as well.
Question: If there are no adjuvants and just the mRNA, why would there be side effects besides pain at the injection site? One participant experienced “fever, chills, muscle and joint aches, really hot hands and feet, headache, general malaise, cough.” Thanks.
The lipid nanoparticle and mRNA are foreign, which is presumably why they also elicit an innate immune response. One of the innate immune responses is inflammation, which causes pain. Please note that I don’t have any first-hand knowledge, but it makes sense that the contents of any injection can cause pain at the injection site, since they are foreign to the body. In terms of fever, chills, joint aches, etc., these are often companions of the acquired immune response. As I understand it (and I am not a health care professional), they are to be expected in people who have a strong acquired immune response or have some other issue that requires the body to divert resources to the immune system.
Thanks for the explanation! The company that I work for provides laboratories for these industries and have even built special virology labs for them this year. It’s good to see how we are contributing.
That’s awesome, Jim!
Might using the body’s cells to make the targeted protein generate a risk of autoimmunity, perhaps to the tissues types of the cells that end up making the protein, since they might be viewed by the immune system as part of “the problem”?
I don’t see how, since cells routinely do this kind of display. Of course, if it happened in the short term, it would show up in the clinical trials. If it is a long-term effect, we won’t know for a while, just as we won’t know the long-term consequences of infection for a while.
Dr. Wile,
Good to know the Pfizer vaccine does not use aborted fetal cell lines. However, there are several vaccines that do use them. They come from two main sources, WI-38 and MRC-5. Apparently there are several other cell lines that have been obtained over the years (see https://bioethics.georgetown.edu/2017/01/is-it-true-that-there-are-vaccines-produced-using-aborted-fetuses/). Also, a more recent line has been developed in 2015 called WALVAX-2.
Sometime ago I spoke with a cattle rancher who reminded me of how Mad-Cow disease started; ground up cow parts were mixed with their feed. There are studies linking the increase in autism with these vaccines (see https://www.soundchoice.org/autism/). In the end, God is not going to honor the creation of vaccines derived from fetal cell lines that have come from abortions, whether they are from 40 years ago or five. It’s a violation of the 6th commandment not to murder. If we change the scenario to killing a baby outside the womb, a child, or an adult, these justifications quickly disappear.
I discuss those vaccines in the article I linked, and I discuss why I (and almost all pro-life groups) think it is ethical to use them. Please read that article. You are free to disagree, of course.
Great article, thanks for the info Dr. Jay,
What I want to know is, from a scientific standpoint, what would be the most effective use of these vaccines:
-Make sure everyone that wants it here in the US gets it, and then share with the rest of the world
or
-Only use enough here in the US to reach “herd immunity” and then share with the rest of the world.
In my humble opinion, I would follow the same common sense logic that is used in flight safety: “Please secure your own oxygen mask before assisting others.” If we don’t first obliterate the virus here in the US, we won’t be of any help to anyone else, especially if, in failing to do so by only reaching “herd immunity,” someone contracts the virus and then brings it to another country.
I would say that these vaccines were produced because of a partnership between the US government and private companies. Thus, US citizens and people associated with the companies should be taken care of first. However, I do think we should help the rest of the world as much as possible.
I completely agree. What I’m wondering though is if the option of helping the rest of the world as much as possible by only reaching “herd immunity” in the US wouldn’t just hurt both the US and the rest of the world in the long run, because, hypothetically, the US didn’t fight the virus aggressively enough, and then an outbreak occurred among the unvaccinated, and then someone carried the virus to another country, and caused an outbreak there.
So my question is, scientifically, would that be a likely scenario if the US tried to vaccinate “Just enough to reach herd immunity,” or could the US plausibly vaccinate “Just enough to reach herd immunity,” and help the rest of the world without a hypothetical outbreak occuring in the US?
I don’t think we would really know ahead of time how many it would take to reach herd immunity. It would depend on population density, etc. Thus, I don’t think that’s a viable strategy.
Dr. Wile, thank you for this clear and understandable explanation. You speak the same way in your texbooks, which is why I have taught our homeschool group with them for the past three years. In fact, we are currently on the cell section in biology which relates to your article so well.
My question or questions are as follows: Since my family and I have had Covid-19, from the literature you have read, is it correct logic that we would not need the vaccine? I am under the impression that Pfizer’s vaccine would be a one time injection-or at least not every year. So following that logic, why is it that experts are saying the I can get Coivd-19 again in 6 months if I have already had it? Wouldn’t that mean that the vaccine would need to be given again after 6 months? Hope my confusion makes sense. Thanks! Liz
I would think there is no need to get the vaccine if you had COVID-19. Your acquired immune system already has the memory, so I don’t think it would help at all. The “experts” saying that you can get it again in 6 months are speculating. The antibodies seem to decay away pretty quickly, but that doesn’t mean you lose immunity, since the memory of your acquired immune system is not in the antibodies. It is in the memory cells that are made. There is nothing I have seen in the literature to indicate that you can catch it again. There have been isolated cases in which doctors say a person has caught it a second time, but that is very hard to confirm.
Thanks for spelling out how this works for us, Dr. Wile!
I’m currently participating in the Jannsen trials and had just enough tenderness in my arm to be hopeful I did not get the placebo.
One of the questions that came up during our orientation was what to do if a vaccine becomes mandatory during the 2 years of our study. The PA I talked to did not anticipate any adverse reaction to having a double dose of the vaccine since the way they function is so different from the dead/inert whole microbial versions.
So, while it may not actually help anything, at least taking a secondary vaccine should not be something harmful to anyone who already has developed immunity.
Thanks for sharing, Cheri. I hadn’t considered that kind of situation.